News
Stay up to date on the latest crypto trends with our expert, in-depth coverage.

1Bitget UEX Daily | Hormuz Reopening Becomes Negotiation Focus; Nvidia Market Cap Breaks $5.2 Trillion; Storage Stocks All Hit New Highs (April 28, 2026)2Alphabet (GOOGL) Q1 2026 Earnings Preview: Cloud Growth Above 50% and AI Monetization in Focus3IOSG In-Depth Analysis of MSTR STRC: The BTC Financing Flywheel Behind 11.5% Yield

Zacks Industry Outlook Highlights Publicis Groupe , Omnicom and Stagwell
Finviz·2026/02/18 09:57

Zacks Industry Outlook Highlights Cisco, Extreme Networks and Radcom
Finviz·2026/02/18 09:54
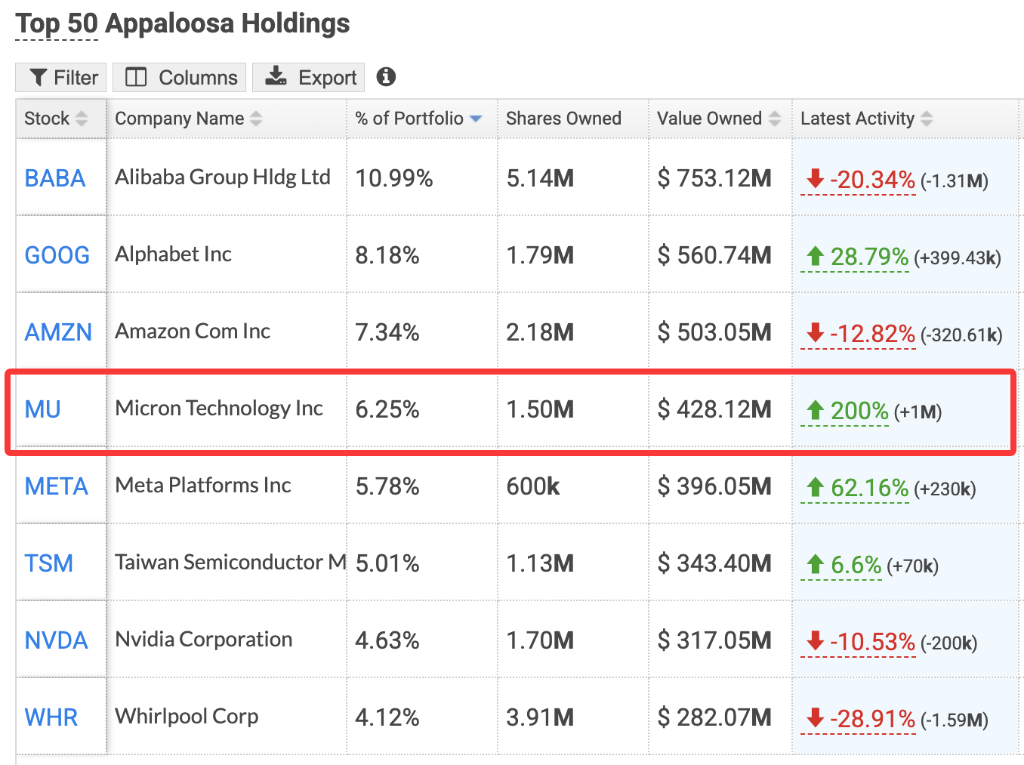
Wall Street Tycoon Tepper Bets on AI: Micron Holdings Surge 200%, Newly Buys Korean ETF
华尔街见闻·2026/02/18 09:53

Jim Farley Says Ford's Universal EV Platform Is Important To 'Win Against China'
Finviz·2026/02/18 09:18


Facing AI Competition Threat, Pinterest Launches "Red Alert" Project
新浪财经·2026/02/18 09:09

xMoney Integrates USDC on Hedera – A New Era for High-Performance Stablecoin Payments
BlockchainReporter·2026/02/18 09:00

Only The Federal Government Has The Power To Regulate Prediction Markets: CFTC
CoinEdition·2026/02/18 08:54
Flash
12:41
In the latest earnings conference call, the CEO of Coca-Cola pointed out that, due to persistent inflation, increased macroeconomic uncertainty, and market volatility caused by conflicts in the Middle East, some consumers are experiencing significant financial pressure.The head of this global beverage giant emphasized that the interplay between geopolitical tensions and the inflationary environment is reshaping global consumption patterns. Although Coca-Cola maintains resilience through flexible pricing strategies and product portfolio optimization, macroeconomic headwinds are indeed constraining consumer purchasing power in some markets. Notably, conflicts in the Middle East have not only triggered energy price fluctuations but have also produced spillover effects on global markets via supply chains and consumer confidence. In the current climate, companies need to pay closer attention to differences in consumer purchasing power across regions and adopt targeted measures to address these challenges.
12:39
UGI Utilities has signed an agreement with Argo Infrastructure Partners to transfer its electric utility assets located in Pennsylvania.This transaction marks an important step in UGI Group's strategic adjustment, with both parties not disclosing specific financial terms.
12:36
Monad Responds to Official Twitter Account Suspension: No Abnormal Activity, Account Expected to Be Restored Soon On April 28, in response to the community's concerns regarding the suspension of Monad's official Twitter account by the X platform, Monad co-founder Keone Hon stated that the account was banned without any prior warning. He suggested that this could be a mistake on the platform's part, emphasizing that the account had not engaged in any abnormal activities or violated API usage policies. The team has reached out to the X platform support through various channels and expects the account to be restored soon.
Trending news
More3 Top Meme Coins to Accumulate Now — DOGE, PEPE, and BONK
In the latest earnings conference call, the CEO of Coca-Cola pointed out that, due to persistent inflation, increased macroeconomic uncertainty, and market volatility caused by conflicts in the Middle East, some consumers are experiencing significant financial pressure.
News